What is a Diagnostic Reference Level?
A Diagnostic Reference Level (DRL) is a dose guidance value used in medical imaging to indicate whether the radiation dose delivered to a patient during a diagnostic procedure is unusually high compared to what is routinely needed to produce a diagnostic-quality image.
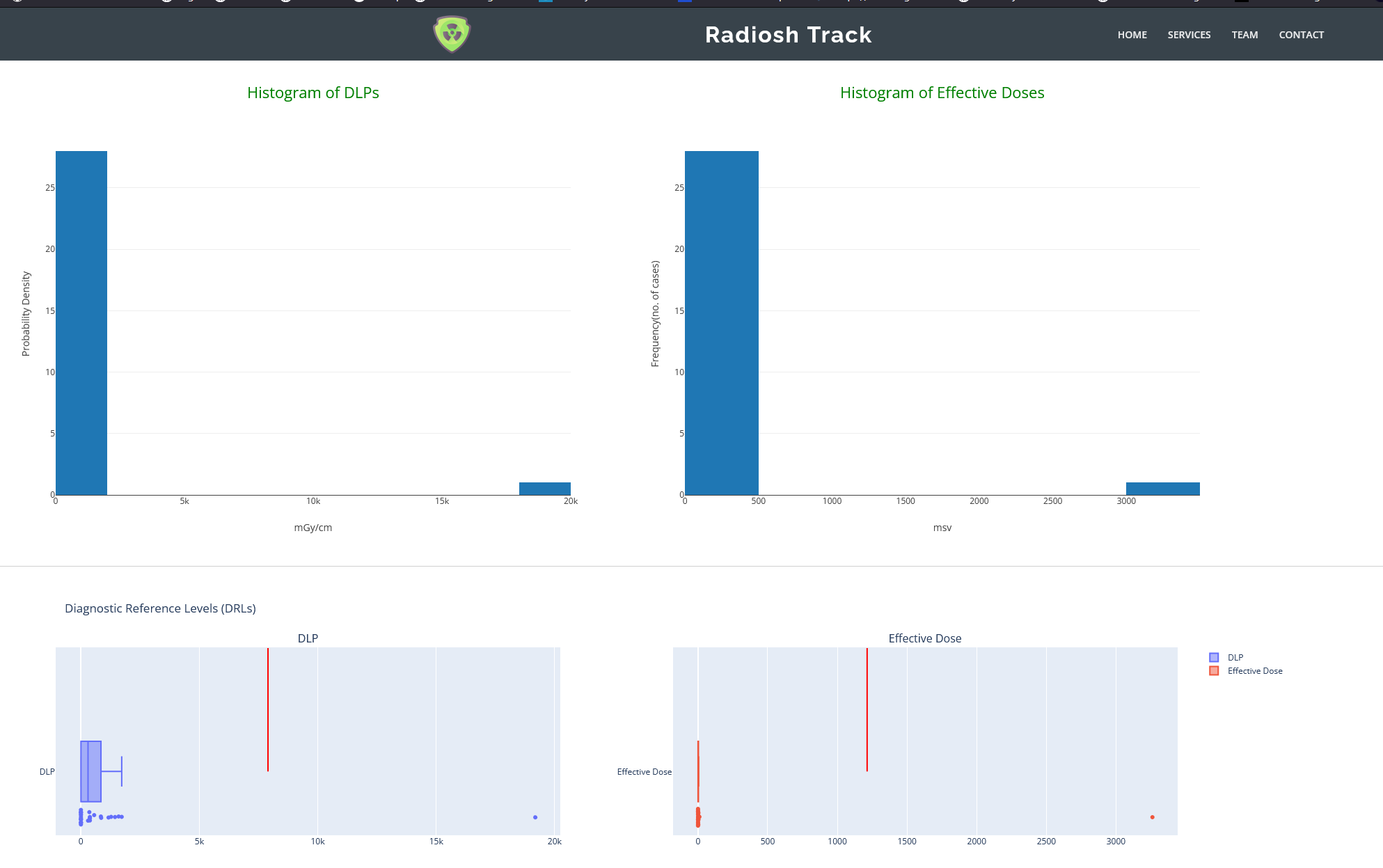
Crucially, a DRL is not a dose limit. It is not a legal threshold that must not be exceeded. It is a prompt for investigation: if a facility's median dose for a given examination consistently exceeds the DRL, it is expected to review its technique, equipment calibration, and protocols to understand why — and to optimise where possible.
DRLs are defined statistically. A national DRL is typically set at the 75th percentile of the dose distribution measured across a large survey of facilities performing the same examination. This means approximately 75% of surveyed facilities are below the DRL, and 25% are above it. The goal of a well-run radiology department is to operate somewhere around the median — not unnecessarily low (which can compromise image quality) and not persistently above the 75th percentile.
Key principle: A DRL exceedance is not a failure — it is a signal. The ICRP and national authorities expect that exceedances will be investigated and, where justified, corrected. The failure is in not investigating.
How DRLs are established
National DRLs
National DRLs are published by regulatory bodies and professional organisations — for example, Public Health England (now UKHSA) in the UK, the European Commission in the EU, and the ACR/NCRP in the United States. They are derived from large periodic surveys of dose data collected from hospitals and imaging centres across the country.
A national DRL survey typically collects dose parameters — such as DAP (Dose-Area Product) for fluoroscopy and X-ray, CTDIvol and DLP for CT, and entrance surface dose for plain radiography — from a representative sample of studies performed at a standard patient size (often defined as a reference patient of 70 kg body weight).
Local DRLs
Beyond national values, local DRLs (also called local reference doses or LDRs) are established within a single department or facility. A local DRL is set at the 75th percentile of the facility's own dose distribution for a given examination, derived from at least 20 recent studies on patients of standard size.
Local DRLs serve two purposes:
- They provide a more relevant benchmark than national values, which represent the average performance of all facilities — including those with older equipment or less optimised protocols.
- They enable departments to track their own improvement over time. If a department successfully optimises a protocol, the local DRL drops, providing a new internal target.
What data do you need to track?
The dose parameters that matter depend on the imaging modality:
- CT: CTDIvol (mGy), DLP (mGy·cm), effective dose (mSv)
- X-Ray / Fluoroscopy: DAP (Gy·cm²), entrance surface dose (mGy), exposure time
- Mammography: Mean glandular dose (MGD)
- Nuclear medicine: Administered activity (MBq)
All of this data is encoded in the DICOM header of every image produced by a modern imaging system. The dose information is stored in DICOM Dose Structured Report (RDSR) objects, or — for older systems — in image-level attributes.
Why manual DRL tracking breaks down
In a small facility performing a few hundred studies per year across one or two modalities, DRL compliance can be managed with spreadsheets. A physicist can export dose data monthly, calculate the 75th percentile, and compare it against published DRLs. This is tedious, but feasible.
In a real department — running 6,000, 10,000, or 30,000 studies per year across CT, X-ray, fluoroscopy, and mammography — the manual approach creates several specific failure modes:
1. Latency
Manual tracking is periodic. Most departments review DRL compliance monthly or quarterly. This means a protocol optimisation problem — perhaps a CT technician accidentally applying an adult protocol to paediatric patients — can run for weeks before it is caught. With automated dose monitoring, the exceedance is flagged the same day.
2. Incomplete data
Manual extraction from PACS or modality consoles is error-prone. Studies get missed. Dose data in free-text fields gets misread. Patient weight adjustments are skipped. The result is a dataset that looks complete but has systematic holes.
3. Audit unreadiness
When a regulatory inspection or accreditation audit requires DRL compliance records, departments using spreadsheets often spend days reconstructing data from disparate sources. Automated systems maintain a continuous, audit-ready record with no reconstruction required.
What good DRL compliance looks like in practice
A department with a mature dose management programme typically has the following in place:
- Automated DICOM ingestion — dose data flows directly from the modality or PACS into the dose management system without manual export.
- Real-time exceedance alerts — when a study's dose exceeds either the national or local DRL, the physicist or department head is notified automatically.
- Monthly or quarterly review meetings — the dose management system generates a report; the team reviews trends, investigates persistent exceedances, and updates local DRLs after protocol optimisations.
- Documented optimisation cycles — when a protocol is changed in response to dose data, the change is recorded with before/after dose distributions. This documentation is invaluable during audits.
The role of automated dose management software
A modern dose management system handles the data pipeline that makes the above workflow possible. It ingests DICOM RDSRs automatically, parses dose parameters for each modality, applies body-size normalisation where relevant, computes running statistics, and flags exceedances — without a physicist needing to touch a spreadsheet.
More advanced systems add natural-language query interfaces, so a physicist can ask questions like "show me all CT head studies last month where CTDIvol exceeded 60 mGy" and receive a filtered table in seconds, rather than writing a database query or manually filtering a spreadsheet column.
The outcome is not just time saved — it is a fundamentally more reliable compliance process. Human review is still essential for interpreting results and deciding on protocol changes, but the data collection and initial flagging no longer depend on human consistency.
ICRP Publication 135 emphasises that dose management systems should be "integrated into routine clinical workflow" rather than treated as a separate, periodic audit exercise. Automation is the practical path to achieving that integration.
Key takeaways
- DRLs are investigation thresholds, not dose limits — exceedances require review, not automatic alarm.
- National DRLs are set at the 75th percentile; local DRLs should be established from your own data.
- Every modern DICOM system encodes dose data — the barrier to automated compliance is software, not hardware.
- Manual tracking creates latency, data gaps, and audit risk that automated systems eliminate.
- The goal is continuous, workflow-integrated dose monitoring — not a monthly spreadsheet exercise.