The state of dose management before AI
For most of the history of digital radiology, dose management has been a manual exercise. A medical physicist periodically exports dose data from a PACS or modality console, loads it into a spreadsheet, computes statistics by hand, compares them against DRL tables from a national guidelines document, and writes a compliance report.
This works. But it is slow, error-prone, and fundamentally reactive. By the time a physicist notices that CT chest doses have been running 40% above the DRL for the past six weeks, several hundred patients have already received that excess dose. The spreadsheet didn't alert anyone — it only confirmed the problem after the fact.
The gap between "dose data exists" and "dose insight is acted upon" is where AI is making its most practical contribution — not by replacing the physicist's clinical judgement, but by eliminating the manual work that delayed it.
What AI actually does well today
Natural language queries over dose data
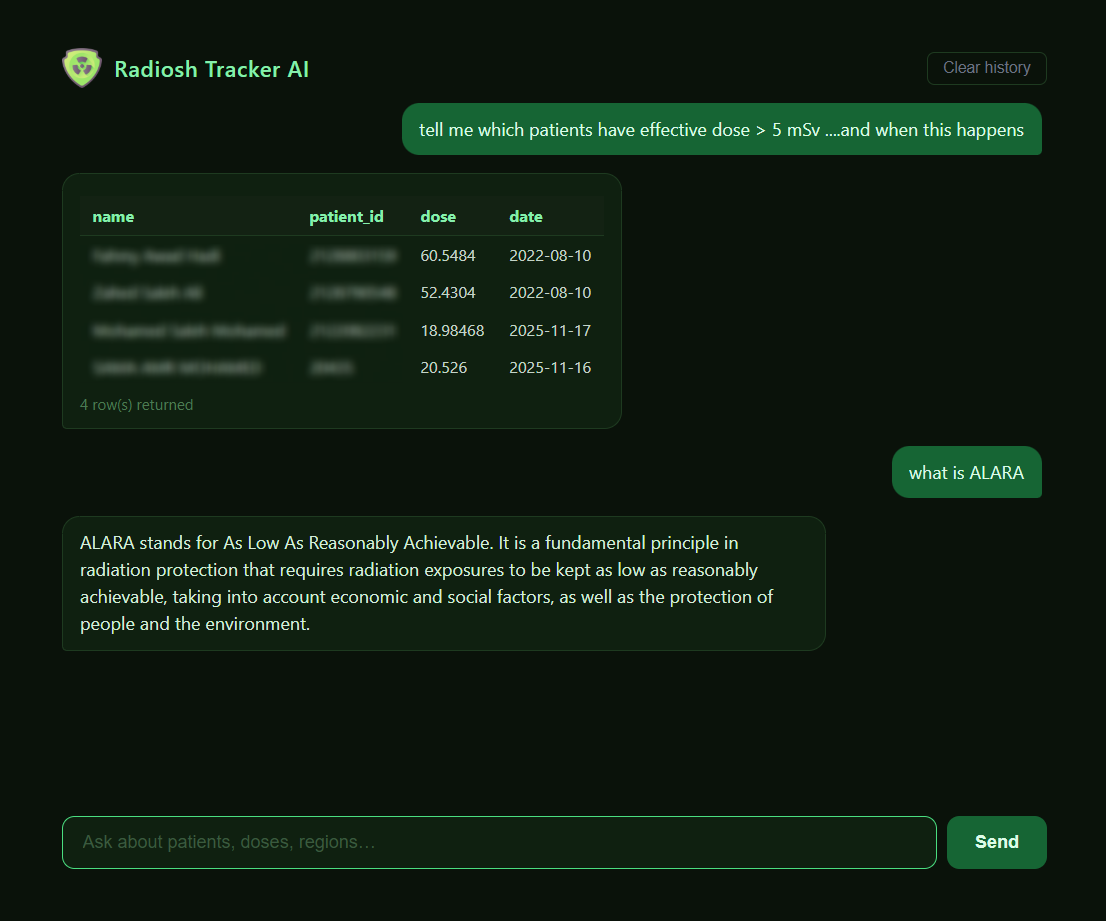
The most immediately useful AI application in dose management is not a complex model — it is a large language model interface on top of a structured dose database. Instead of writing a SQL query or filtering a pivot table, a physicist can type a question in plain English and receive a structured answer.
| Patient ID | Dose (mSv) | Date |
|---|---|---|
| PT-00142 | 60.5 | 2022-08-10 |
| PT-00198 | 52.4 | 2022-08-10 |
| PT-00371 | 18.9 | 2025-11-17 |
| PT-00412 | 20.5 | 2025-11-16 |
This is not a trivial capability. A physicist who previously spent 20 minutes filtering a spreadsheet to answer a straightforward compliance question can now get that answer in ten seconds — and follow it with a contextual question about what the result means, without switching applications or opening a textbook.
Automated exceedance detection
Rule-based exceedance detection has existed in dose management software for years — if a dose value exceeds a threshold, raise a flag. AI improves on this in two ways:
- Pattern detection across dimensions — rather than flagging individual study exceedances, an AI-assisted system can identify that doses have been trending upward across a specific technologist's shift, or that one CT suite is consistently producing higher doses than an equivalent suite in the same department, without either individually exceeding the DRL threshold.
- Anomaly detection in unlabelled data — some dose anomalies don't have a pre-defined threshold to trigger against. A study where the DAP value was recorded correctly but the body part is inconsistently coded may be silently skipped by rule-based systems. Statistical anomaly detection catches distributions that look wrong even without a named rule.
Automated report generation
Generating a monthly DRL compliance report manually typically takes a medical physicist two to four hours. It involves querying the database, computing statistics, building charts, formatting tables, writing interpretation text, and producing a document suitable for the department's quality records.
AI-assisted report generation compresses this to under two minutes. The system queries its own database, computes the relevant statistics, identifies which modalities are above or below DRL thresholds, generates narrative interpretation (e.g. "CT chest doses have been trending downward since the protocol update in March"), and produces a formatted PDF ready for signature.
The physicist's role shifts from report author to report reviewer — a fundamentally more valuable use of their expertise.
What AI cannot do — and shouldn't be asked to
A candid assessment of current AI in dose management requires acknowledging its real limitations.
✓ AI does well
- Querying and filtering structured dose data
- Detecting statistical anomalies in dose distributions
- Generating formatted compliance reports
- Explaining clinical concepts (ALARA, DRLs, dose units)
- Tracking trends over time across large datasets
⚠ Requires clinical judgement
- Deciding whether a dose exceedance was clinically justified
- Recommending specific protocol parameter changes
- Interpreting dose in the context of a specific patient's diagnosis
- Replacing the medical physicist's review and sign-off
- Making dose-reduction decisions for complex or paediatric cases
The right mental model for AI in dose management is a highly capable assistant, not an autonomous decision-maker. It eliminates the time a physicist spends on mechanical tasks — data extraction, computation, formatting — freeing them to spend more time on the tasks that actually require their training: protocol design, clinical interpretation, and optimisation decisions.
On AI hallucination in clinical contexts: Language models can produce plausible-sounding but incorrect answers, particularly when queried about specific patient data they were not trained on. In a dose management system, this risk is mitigated by grounding AI responses directly in the facility's own structured database — the AI is querying real data, not generating from training memory. Users should still verify anomalous results against the raw data before acting on them.
What's coming next
The near-term development trajectory for AI in dose management points in three directions:
Protocol recommendation
Given a patient's body habitus, clinical indication, and the department's own dose-quality history, an AI system could suggest starting protocol parameters — not to replace the physicist's final decision, but to give them a data-informed starting point. This is particularly useful when onboarding a new modality or adapting protocols for a patient population with different characteristics than the department's historical baseline.
Cross-facility benchmarking
Aggregated, de-identified dose data from multiple facilities enables AI-driven benchmarking that goes beyond national DRLs. Instead of comparing against a national 75th percentile set from a survey conducted several years ago, a department could compare against a real-time peer group of similar-sized facilities using similar equipment. This is a significantly more relevant and actionable benchmark.
Predictive dose alerting
Rather than detecting exceedances after they occur, predictive models could flag that a department is on a trajectory toward a DRL exceedance before it happens — based on trends in scanner output, patient population changes, or seasonal workload variation. This shifts dose management from reactive to genuinely proactive.
Key takeaways
- AI's most immediate value in dose management is eliminating manual data work — query time, report generation, threshold monitoring — so physicists can focus on interpretation and decisions.
- Natural language interfaces make dose databases accessible to clinical staff without database expertise, broadening who can ask questions of the data.
- Current AI cannot replace clinical judgement on protocol decisions, justified exceedances, or patient-specific dose optimisation — and it shouldn't try to.
- The grounding of AI responses in structured facility data, rather than general training memory, is essential for clinical reliability.
- The near-term frontier is protocol recommendation, cross-facility benchmarking, and predictive alerting — all of which require accumulated dose data as their foundation.